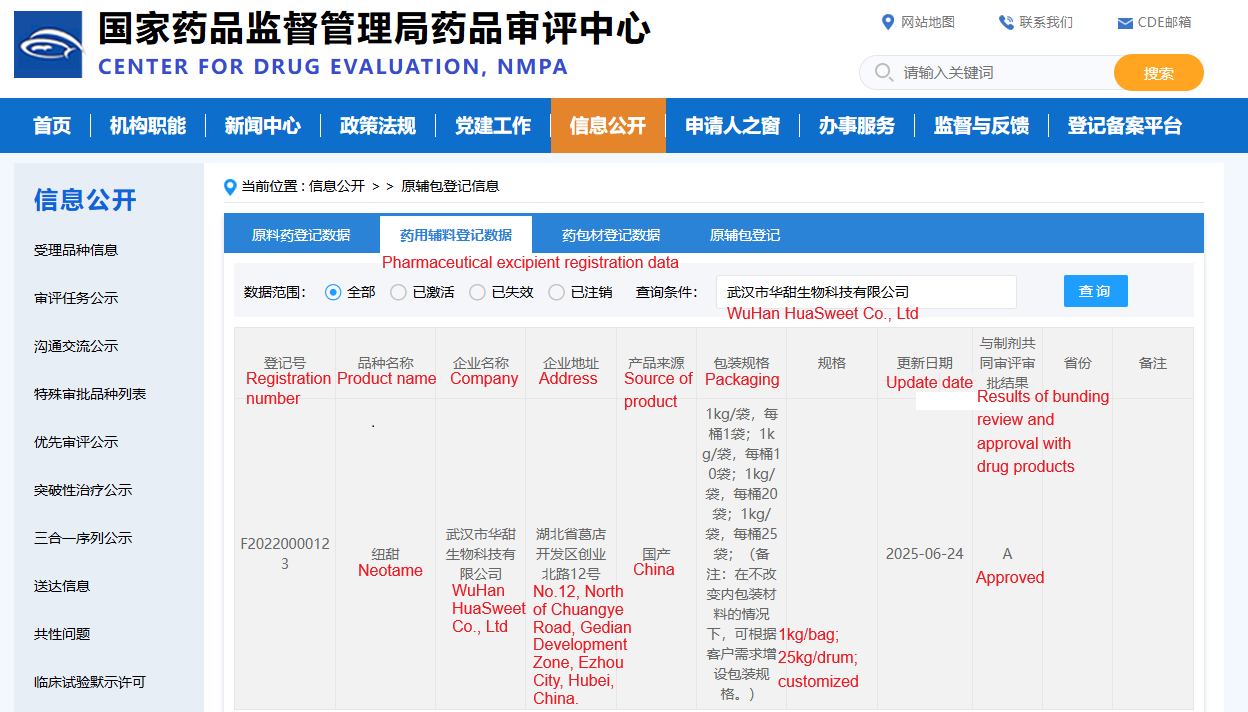
On June 24, 2025, CDE (the Center for Drug Evaluation under the National Medical Products Administration in China) officially approved HuaSweet pharmaceutical-grade neotame.
Wuhan HuaSweet, in collaboration with its partners, provided a comprehensive set of data, including neotame's physical and chemical properties, toxicology data, metabolic analysis, and application results. After three years of rigorous review by various CDE departments, Huatian successfully received approval, becoming the first manufacturer in China to receive pharmaceutical-grade neotame approval. Neotame, used as a flavoring agent in pharmaceuticals, has earned high recognition from pharmaceutical companies for its high sweetness, minimal dosage, excellent efficacy, and safety.
From formulating the national standard for neotame, becoming a USP supplier, obtaining core patent authorization, promoting market application, being selected as a National-level specialized and innovative enterprise to the approval of pharmaceutical-grade neotame, all these fully demonstrate that Wuhan HuaSweet has always been leading the development of the neotame industry.